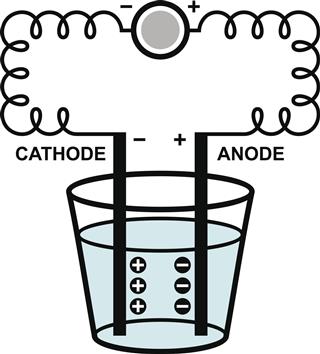
A comparative study of what ionic compounds and molecular compounds are will help you understand the differences between the two of them.
A compound is formed when two or more elements go on to combine together to form a new substance. The chemical and physical properties of the compound that is eventually formed are different from those of the constituent elements. Most of the substances found in nature exist in the form of compounds, rather than as free elements. There are less than a hundred different elements that can be found in nature, but the compounds that they form are actually found in millions.
Let’s have a look at what ionic and molecular compounds are, their various properties, and the main points of difference between them.
Ionic Compounds
When an atom loses or gains one or more electrons, it becomes positively or negatively charged, and this charged atom is called an ion. Ions are unstable, and hence, a negatively charged ion gets attracted towards a positively charged ion to form an ionic compound. The bonds that they form are called ionic bonds. If an atom loses an electron, it acquires a positive charge, and if it gains an electron, it acquires a negative charge.
A common example of an ionic compound is common salt or NaCl, in which the sodium atom gains one pair of electrons from the chlorine atom, and forms an ionic bond. Other compounds like NaBr (sodium bromide), KCl (potassium chloride), and CaCl2 (calcium chloride) are also ionic compounds.
Molecular Compounds
Molecular compounds are also called covalent compounds. In these compounds, the atoms of the elements share one or more electrons between them. The bonds that are formed as a result of the sharing of electrons are called covalent bonds. Equal sharing of electrons between the atoms results into non-polar bonding, whereas unequal sharing results into polar bonds.
A common example of a molecular compound is water or H2O, in which the hydrogen and oxygen atoms share a pair of electrons between them, thus forming covalent bonds. Other compounds like HCl (hydrogen chloride), CH4(methane), and CO2 (carbon dioxide) are also molecular compounds.
Properties of Ionic and Molecular Compounds
Structure
Ionic Compounds: Ionic compounds are usually in the solid state at standard temperature and pressure (STP). This is due to the strong attraction between their positively charged and negatively charged ions. They form a crystalline structure called crystal lattice.
Molecular Compounds: They usually exist in the liquid or gaseous state at standard temperature and pressure. This is because of a weak force of attraction between the atoms forming covalent bonds.
Melting and Boiling Points
Ionic Compounds: Due to the strong attractive forces between the positive and negative ions in an ionic compound, a lot of energy is required to break the ionic bonds between the oppositely charged ions. So they have a high melting and boiling point.
Molecular Compounds: Due to a weak force of attraction between the atoms, these compounds usually have a low melting and boiling point.
Hardness
Ionic Compounds: Ionic compounds are very hard because of their closely-packed lattice structure and the ionic bonds between the charged ions. They are very brittle, and it is difficult to bend them.
Molecular Compounds: The solid forms of molecular compounds are generally very soft and brittle. The covalent bonds in such compounds are flexible, and bend or break easily.
Electrical Conduction
Ionic Compounds: In their solid state, ionic compounds do not conduct electricity. But when melted in an aqueous solution, they act as strong electrolytes and conduct electricity.
Molecular Compounds: They are poor conductors of electricity and heat. When they dissolve in an aqueous solution, they still remain in their molecular form. Due to lack of free ions, they do not carry electric charge very well, and thus, act as poor conductors of electricity.
Solubility
Ionic Compounds: Most ionic compounds are soluble in water, and insoluble in non-polar liquids and organic compounds.
Molecular Compounds: Usually, molecular compounds are insoluble in water, but are soluble in organic solvents.
Differences between Ionic and Molecular Compounds
- An ionic compound is formed by the reaction of a metal with a non-metal, whereas a molecular compound is usually formed by the reaction of two or more non-metals.
- In ionic compounds, the ions are held together due to electrical attraction, whereas, in molecular compounds, the atoms are held together by the attraction between the atoms due to shared electrons.
- Ionic compounds are charged ions, whereas molecular compounds consist of molecules.
- A molecular compound cannot conduct electricity in any state, whereas an ionic compound, when dissolved in an aqueous solution, can act as a good conductor of electricity.
- Ionic compounds are more reactive than molecular compounds.
- The enthalpy of fusion (heat energy absorbed when a solid melts) and enthalpy of vaporization (heat energy absorbed when a liquid boils) are higher in ionic compounds.
- Molecular compounds are more flammable than ionic compounds.
- Molecular compounds are softer and more flexible as compared to ionic compounds.
- Ionic compounds have a higher melting and boiling point than molecular compounds.
The above differences also help distinguish and identify whether a given unknown compound is ionic or molecular, by conducting appropriate tests in a lab, and finding out the differences in chemical and physical properties of the compound.