Butyl acetate is a clear-liquid chemical used in various manufacturing industries. Read this ScienceStruck article to know a lot more about its properties and various uses.
Did You Know?
Butyl acetate becomes toxic at high concentrations of more than 950 mg/m3.
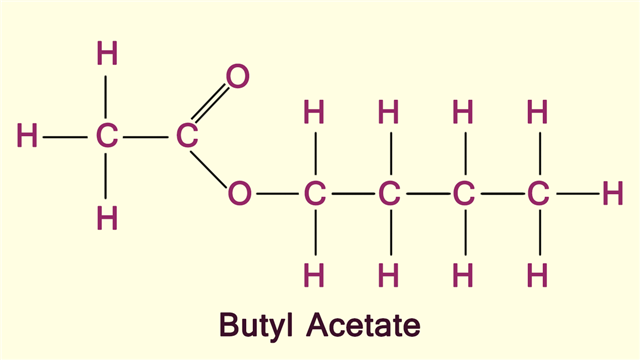
Butyl acetate is an organic compound which occurs naturally in food products. However, it can also be produced chemically. It is commonly used as a solvent in the production of lacquers and paints. It is a colorless liquid with a low viscosity, having a sweet banana-like odor. This liquid is volatile but flammable in nature.
In the following sections, we’ll have a look at its structure, formation, properties, and uses.
Butyl Acetate Chemical Structure
Formation
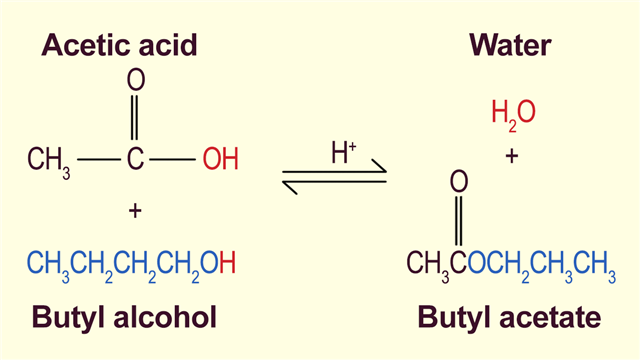
Butyl acetate is a type of an ester; a substance formed by the reaction between an acid and an alcohol along with the elimination of water. In this case, the reaction is between acetic acid and butyl alcohol.
Butyl acetate has four isomers: isobutyl acetate, sec-butyl acetate, tert-butyl acetate, and n-butyl acetate. An isomer is a compound that exists in forms having different arrangements of atoms, but the same molecular weight. The following table provides information regarding the properties of all four isomers of butyl acetate.
Properties
| Property | isobutyl acetate | sec-butyl acetate | tert-butyl acetate | n-butyl acetate |
| Appearance | colorless liquid | colorless liquid | colorless liquid | colorless liquid |
| IUPAC Name | isobutyl acetate | sec-butyl acetate | tert-butyl acetate | butyl acetate |
| Systematic Name | 2-methylpropyl acetate | 1-methylpropyl | 1,1-dimethylethyl | butyl ethanoate |
| Other Names | isobutyl ester | sec-butyl ester; s-butyl acetate; 2-butyl acetate | acetic acid tert-butyl ester; t-butyl acetate | n-butyl acetate; 1-butyl acetate; acetic acid n-butyl ester; n-butyl ethanoate; butyl ester of acetic acid; 1-acetoxybutane; 1-butanol, acetate; butyl ester, acetic acid |
| Formula | C6H12O2 | C6H12O2 | C6H12O2 | C6H12O2 |
| Molar Mass | 116.16 g/mol | 116.16 g/mol | 116.16 g/mol | 116.16 g/mol |
| Density | 0.875 g/cm3 | 0.87 g/cm3 | 0.8593 g/cm3 | 0.8825 g/cm3 |
| Melting Point | −99°C (−146°F; 174 K) | −99°C (−146°F; 174 K) | −62°C (-79.6°F; 211.15 K) | −78°C (−108°F; 195 K) |
| Boiling Point | 118°C (244°F; 391 K) | 112°C (234°F; 385 K) | 97.8°C (208.0°F; 370.9 K) | 126.1°C (259.0°F; 399.2 K) |
| Flash Point | 17.77°C (64°F; 290.92 K) | 31°C (88°F; 304.15 K) | 11°C (51.8°F; 284.15 K) | 22°C (72°F; 295 K) |
| Specific Gravity at 20/20°C | 0.873 | 0.872 | 0.87 | 0.8826 |
| Solubility in Water | 7 g/L (20ºC) | 0.8 g/100 mL (20°C) | insoluble | 0.68 g/100 mL (20°C) |
| Vapor Pressure | 15 mmHg (20°C) | 10 mmHg (20°C) | 47 mmHg (25°C) | 11.25 mmHg (25°C) |
| CAS Number | 110-19-0 | 105-46-4 | 540-88-5 | 123-86-4 |
Uses
● tert-Butyl acetate is used as an additive to gasoline to prevent engine knocking. It is also used as a solvent for lacquers.
● n-Butyl acetate easily dissolves nitrocellulose, a nitric acid ester used in lacquers. Therefore, it is used as a solvent in the manufacture of nitrocellulose lacquers.
● It is used as a solvent in the production of nail polish, nail polish removers, basecoats, and other nail-care products.
● It is used as a synthetic-fruit flavoring in foods such as candy, ice cream, baked goods, cheeses, yogurts, etc.
● It is used in as a solvent in organic chemistry laboratories.
● It is used in the manufacture of Penicillin-G, the principal component of many commercial antibiotics.
● It is used in the manufacture of artificial leather, cleaners, photographic films, plastics, and safety glass.
● Due to its low solubility in water, n-Butyl acetate is used as an extractant in the manufacture of pharmaceutical preparations.
● It is used as a diluting agent for dyes in inks used for marking vegetables and fruits.
● n-butyl and isobutyl acetate both are used in the production of perfumes.
● sec-Butyl acetate is used as a solvent for nail polish and lacquer. It is also used in the production of paper coatings.
Butyl acetate is known to cause respiratory problems. Hence, prolonged exposure to high concentrations should be avoided to prevent any sort of damage to the nervous system.