If you are looking for examples of endothermic reactions in everyday life, this article has just what you are looking for. Here, we explain what these reactions are and present examples of the same.
One of the most elusive concepts to grasp in physics is the nature of energy. Energy manifests itself in various forms and we study its properties indirectly through the changes it brings about in matter. All types of chemical reactions and physical changes can be broadly classified into two categories, which include endothermic and exothermic reactions. In chemistry or physics, the basis for this classification is the input or output of energy, which accompanies any reaction.
Definition
Any chemical reaction that requires an external input of energy, in the form of heat, from the surroundings, is an endothermic reaction. So these reactions are accompanied by absorption of heat from the surroundings, which brings down the external temperature.
Thermodynamics is devoted to the study of energy exchanges that occur through any chemical reaction and how they affect the state variables of any system. If any endothermic process occurs under conditions of constant pressure, there is an increase in the enthalpy of the system and if it occurs at constant volume, there is an increase in the internal energy of the system. One way of remembering the meaning of the term – ‘Endothermic’ is to break it down into two parts. ‘Endo’ means inside, while ‘thermic’ refers to heat.
Endothermic and exothermic reactions are exact opposites of each other. While the former is triggered by absorption of heat, the latter is accompanied by emission of energy.
Examples
There is a single feature which may be used to identify any endothermic reaction and that is the absorption of heat. Here are some examples from everyday life.
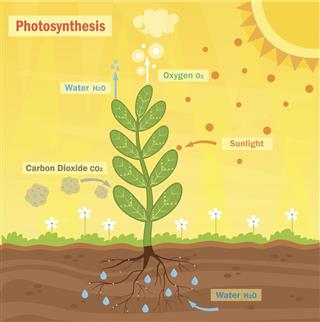
Photosynthesis
The whole food pyramid on Earth is sustained by the presence of photosynthesizing plants that occur at the bottom of this pyramid. It is plants that create ‘food’ in the form of glucose, through the process of photosynthesis. They receive energy in the form of sunlight falling on the leaves, which powers the photosynthesis reaction. Without sunlight, there can be no photosynthesis, which marks this reaction as an endothermic one.
Water Evaporation
When water evaporates and gets transformed into steam, it is because of the heat it receives from the surroundings. As heat energy is received by every water molecule, its vibration energy increases to the point where it can move around freely, creating steam. This heat input from external sources makes it an endothermic reaction.
Melting Ice
Melting of ice is also an example of an endothermic reaction. The heat energy received by ice, from its surroundings, makes it melt into water. Since this physical change cannot occur without the heat input from surroundings, this phenomenon is classified as an endothermic process.
Electrolysis Process
Electrolysis is the decomposition of a molecule into its constituent ions because of the passage of an electric current. The energy required for it, is supplied externally and hence it is another example.
Just remember that when heat is absorbed from the surroundings, in a chemical reaction, it’s an endothermic process.